Mastering the Percent Yield Equation
📋 In this guide
The percent yield equation is a crucial concept in chemistry that allows scientists to determine the efficiency of a chemical reaction. Students often find themselves grappling with this topic because it involves both theoretical understanding and practical application. The percent yield equation provides insight into the actual versus theoretical outcomes of chemical reactions, which is essential for both academic studies and real-world applications.
Understanding the percent yield equation involves more than just plugging numbers into a formula. It requires a comprehension of the underlying principles of chemical reactions and the ability to perform algebraic manipulations accurately. Students often struggle with this because it combines elements of chemistry and algebra, making it a challenging topic for those not well-versed in both subjects. However, mastering this concept can significantly enhance a student's ability to solve complex problems in chemistry.
In this guide, we'll break down the percent yield equation, explaining it in simple terms and providing step-by-step instructions on how to apply it. We'll also explore common mistakes to avoid, real-world applications, and answer frequently asked questions. By the end of this article, you'll have a thorough understanding of what the percent yield equation is, how to calculate it, and why it's important.
Step-by-Step: How to Solve Percent Yield Equation
Step 1: Understanding Actual and Theoretical Yields
Before you can use the percent yield equation, it's important to understand the difference between actual and theoretical yields. The theoretical yield is the amount of product that would be produced in a perfect world where everything reacts completely without any loss, based on stoichiometric calculations. In contrast, the actual yield is the amount of product you actually get from the experiment, which is often less due to factors like incomplete reactions or experimental errors.
Step 2: Calculating Theoretical Yield
To calculate the theoretical yield, you need to perform stoichiometric calculations. These involve using balanced chemical equations to convert moles of reactants to moles of products, and then converting moles of products to grams if necessary. This step requires a solid understanding of mole-to-mole ratios and the ability to perform conversions between moles and grams using molar masses.
Step 3: Measuring Actual Yield
The actual yield is typically measured by weighing the product obtained after the reaction. It's important to ensure that the product is pure and free from contaminants, as impurities can affect the weight and lead to inaccurate calculations. Make sure to use precise measuring tools and follow laboratory protocols to obtain an accurate measurement.
Step 4: Applying the Percent Yield Equation
Once you have both the actual and theoretical yields, you can apply the percent yield equation. Divide the actual yield by the theoretical yield and multiply the result by 100 to get the percent yield. This percentage will give you an idea of how efficiently the reaction was carried out and how much of the starting material was converted into the desired product.
🤖 Stuck on a math problem?
Take a screenshot and let our AI solve it step-by-step in seconds
⚡ Try MathSolver Free →Worked Examples
Example 1
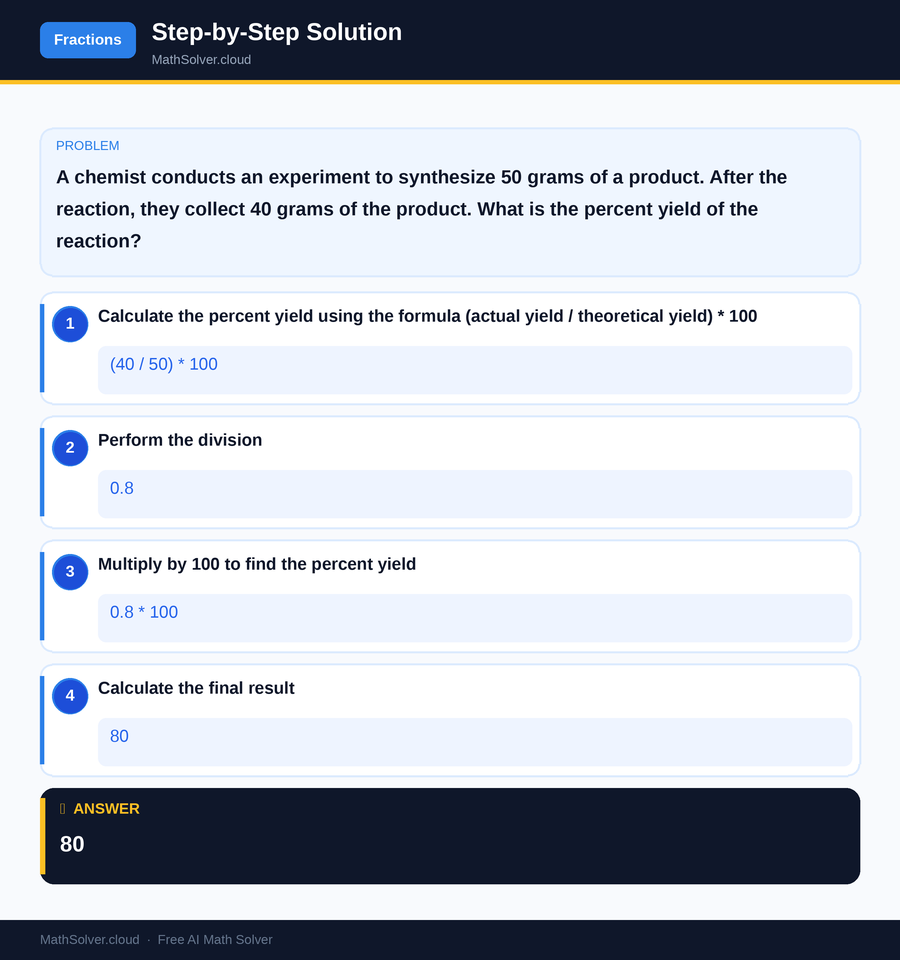
MathSolver Chrome extension solving this problem step-by-step
Example 2
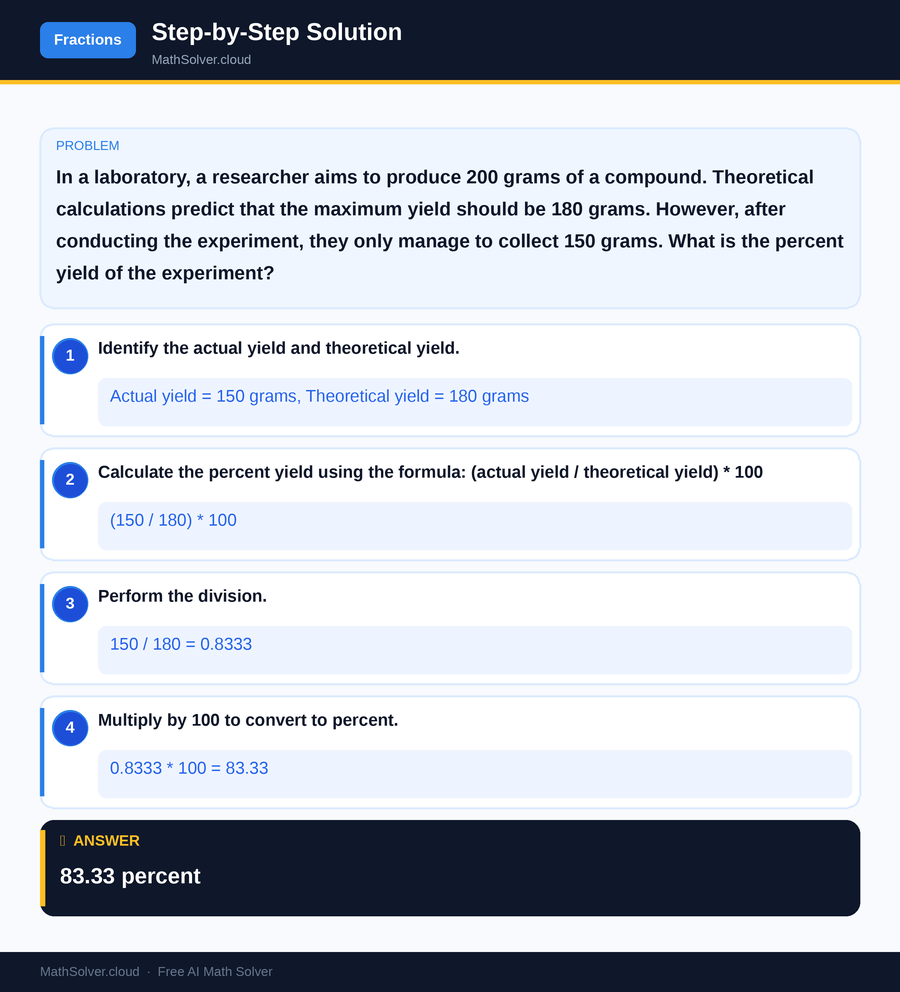
MathSolver Chrome extension solving this problem step-by-step
Common Mistakes to Avoid
One common mistake students make is confusing the actual yield with the theoretical yield when using the percent yield equation. It's important to remember that the theoretical yield is based on ideal conditions, while the actual yield is what is actually produced. Mixing these up can lead to incorrect calculations.
Another frequent error is not considering all reactants and products involved in the reaction. Sometimes, students may overlook side reactions or impurities that can affect the actual yield. To avoid this, always double-check your stoichiometric calculations and ensure that all factors are considered.
Real-World Applications
The percent yield equation is not just a theoretical concept; it has significant real-world applications, particularly in the chemical industry. For instance, manufacturers use percent yield calculations to determine the efficiency of their production processes and to optimize the use of raw materials.
In pharmaceuticals, the percent yield equation is crucial for ensuring that drug production is efficient and cost-effective. By maximizing the yield, companies can reduce waste and improve the sustainability of their operations, ultimately leading to lower costs and more affordable medications.
Frequently Asked Questions
❓ How do you use the percent yield equation in chemistry?
❓ Why is my percent yield over 100%?
❓ How can AI help with the percent yield equation?
❓ What is the difference between percent yield and yield strength?
❓ How is the percent yield equation used in quality control?
🚀 Solve any math problem instantly
2,000+ students use MathSolver every day — join them for free
📥 Add to Chrome — It's Free