Demystifying the Percent Error Equation
📋 In this guide
The percent error equation is a valuable tool in mathematics and science for measuring the accuracy of an experimental value compared to a known or accepted value. This equation helps students and professionals alike determine how accurate their measurements or calculations are. Unfortunately, many students struggle with understanding and applying the percent error equation due to its seemingly abstract nature and the challenge of interpreting the results in a meaningful way. Throughout this article, we will demystify the percent error equation, providing you with a clear understanding of how to apply it effectively.
By learning about the percent error equation, you will gain the ability to assess the reliability of your experimental results. This skill is not just useful in academic settings; it also has practical applications in various fields such as engineering, chemistry, and physics. Understanding this concept will give you a better grasp of the importance of precision and accuracy in scientific measurements.
In this comprehensive guide, we will walk you through the key formula, provide a step-by-step process for applying it, and solve some example problems to cement your understanding. Additionally, we'll highlight common mistakes to avoid, explore real-world applications, and answer frequently asked questions. By the end of this article, you'll feel confident in your ability to use the percent error equation effectively in both academic and real-world contexts.
Step-by-Step: How to Solve Percent Error Equation
Step 1: Identify the Experimental and Actual Values
To apply the percent error equation, the first step is to determine the experimental value and the actual value. The experimental value is the measurement or estimation you have obtained through experiments or calculations. The actual value, on the other hand, is the known or accepted value that you are comparing against. This could come from a trusted source or a theoretically calculated value. Clearly identifying these values is crucial as it sets the foundation for the rest of the calculation.
Step 2: Calculate the Difference
Once you have identified the experimental and actual values, the next step is to calculate the difference between them. Subtract the actual value from the experimental value, and take the absolute value of the result. This step is essential because it gives you the raw error amount, which is the deviation of your experimental result from the actual value. The absolute value ensures that this error is expressed as a positive number, highlighting its magnitude without concern for whether the experimental value is above or below the actual value.
Step 3: Divide by the Actual Value
After calculating the absolute difference, the next step is to divide this difference by the actual value. This division gives you the relative error, which provides a sense of how significant the error is in relation to the actual value. This step is crucial because it normalizes the error, allowing you to compare errors across different scales and magnitudes of measurement.
Step 4: Convert to Percentage
Finally, to convert the relative error into a percent error, multiply the result from Step 3 by 100. This conversion expresses the error as a percentage of the actual value, making it easier to interpret and communicate. A smaller percent error indicates a more accurate measurement, while a larger percent error suggests a greater deviation from the true value. Understanding this final result helps in assessing the reliability of your experiments or calculations.
🤖 Stuck on a math problem?
Take a screenshot and let our AI solve it step-by-step in seconds
⚡ Try MathSolver Free →Worked Examples
Example 1
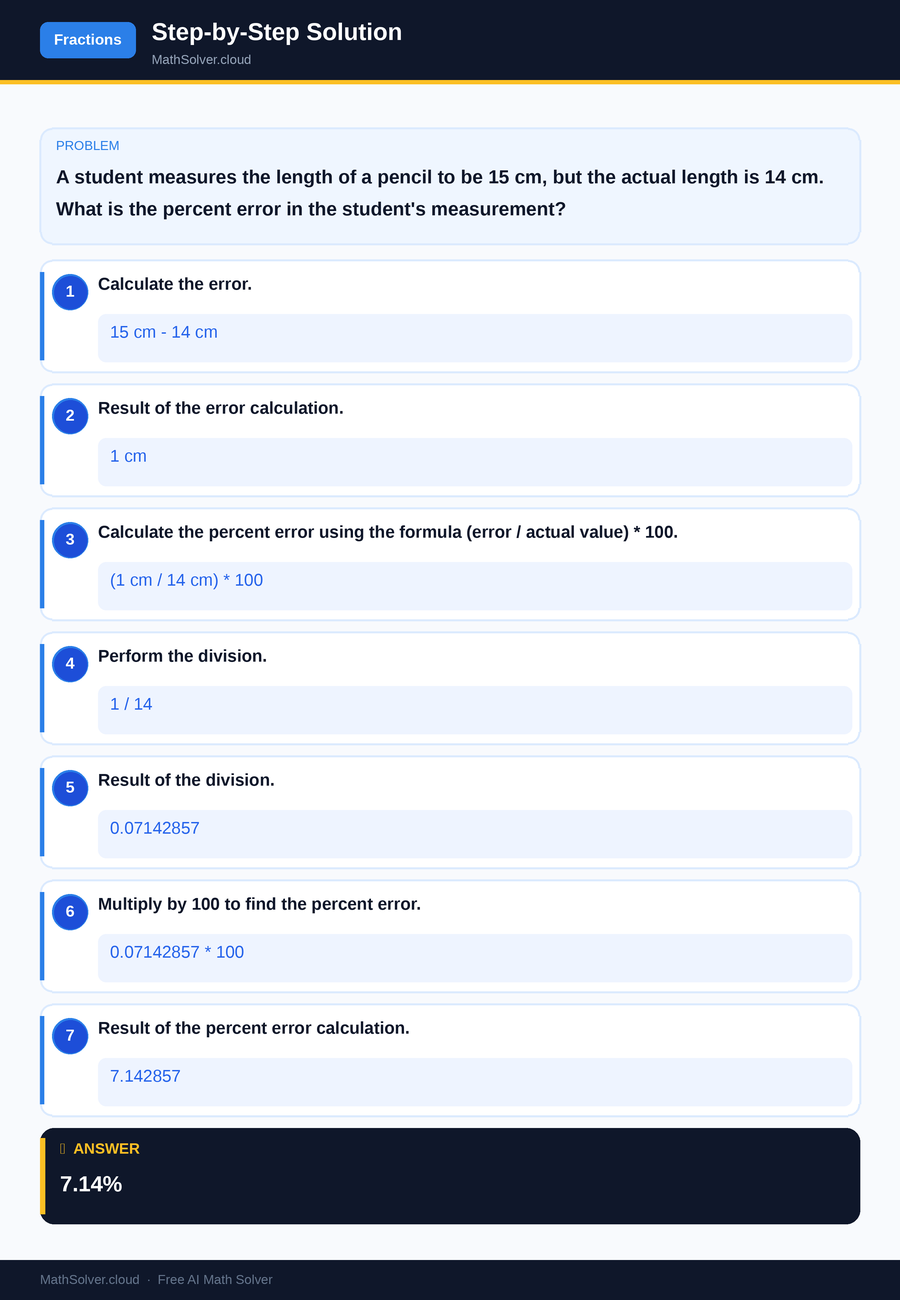
MathSolver Chrome extension solving this problem step-by-step
Example 2
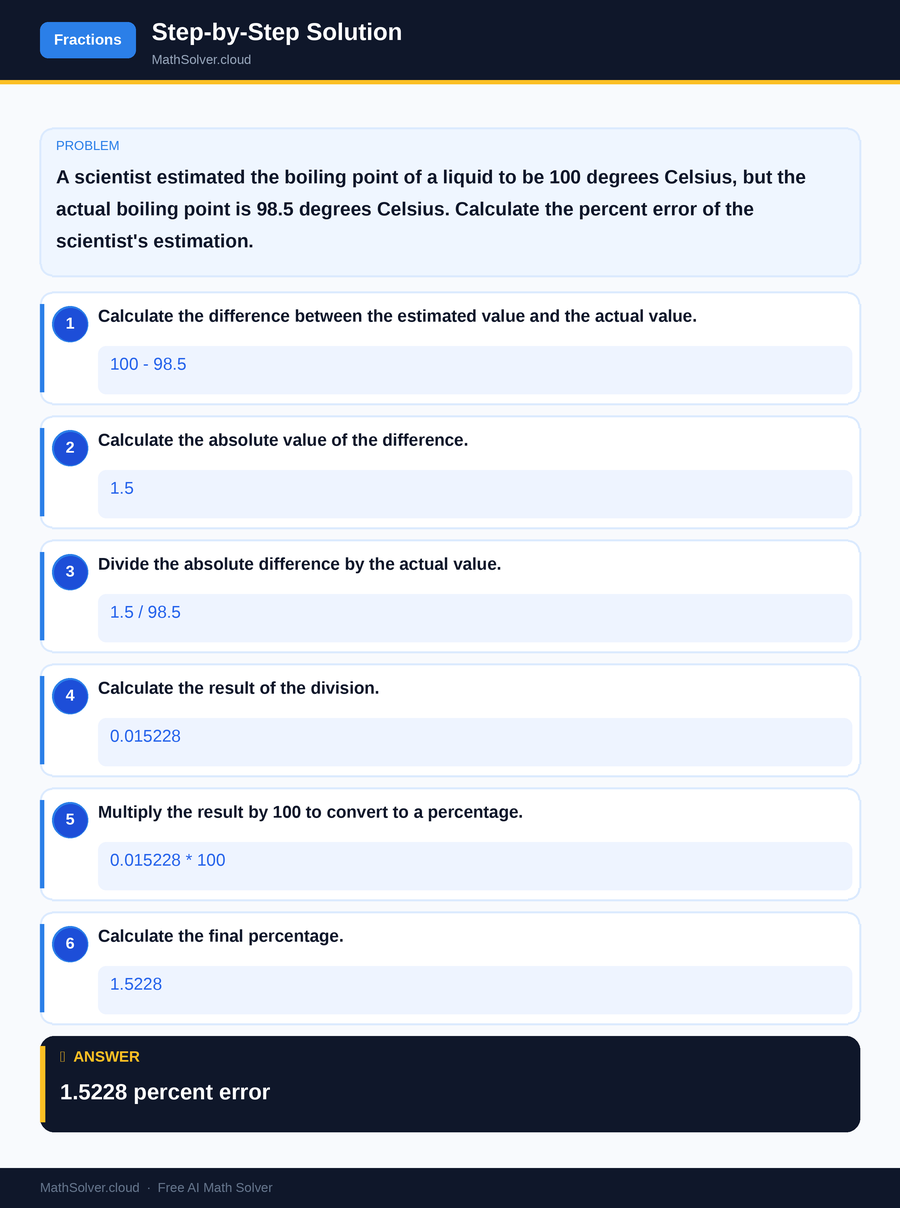
MathSolver Chrome extension solving this problem step-by-step
Common Mistakes to Avoid
One common mistake students make is forgetting to take the absolute value of the difference between the experimental and actual values. This oversight can lead to negative percent errors, which are not meaningful in this context. Always remember to use the absolute value to ensure the error is expressed positively, reflecting its magnitude.
Another frequent error is incorrectly identifying the experimental and actual values. Mixing these values up can result in a completely incorrect calculation. Always double-check which value is experimental and which is the actual value before proceeding with your calculations. By being diligent in these initial steps, you can avoid simple yet impactful errors.
Real-World Applications
The percent error equation is widely used in scientific research and industry to assess the accuracy of experimental results. In fields like chemistry and physics, precise measurements are critical, and percent error helps researchers evaluate the reliability of their data. For example, in chemical reactions, percent error is used to determine the accuracy of yield measurements compared to theoretical predictions.
In engineering, percent error is essential for quality control and ensuring that products meet specified standards. Engineers use this equation to compare measured dimensions or material properties with design specifications, allowing them to identify and rectify deviations efficiently. By understanding percent error, professionals can make informed decisions that impact safety, quality, and performance.
Frequently Asked Questions
❓ What is the percent error equation, and why is it important?
❓ How does the percent error equation differ from other algebra equations?
❓ How can AI help with percent error equation calculations?
❓ When should I use the percent error equation instead of other equations like the quadratic or kinematic equations?
❓ Can percent error be applied in evaluating hypotheses in scientific research?
🚀 Solve any math problem instantly
2,000+ students use MathSolver every day — join them for free
📥 Add to Chrome — It's Free