Mastering the Mass Percent Equation
📋 In this guide
The mass percent equation is a fundamental concept in chemistry and mathematics that helps us understand the composition of mixtures and solutions. It is particularly useful when dealing with solutions, alloys, or any scenario where you need to determine the concentration of a component relative to the whole. Despite its apparent simplicity, students often find themselves confused by the mass percent equation, mainly because it requires careful attention to detail and a strong grasp of basic algebra equations. In this article, we'll break down the mass percent equation, walk you through a step-by-step guide, and provide practical examples to ensure you have a clear understanding of this essential concept.
Understanding the mass percent equation is crucial for students studying chemistry, as it forms the basis for more advanced topics such as molarity and stoichiometry. Many students struggle with this equation because they overlook the importance of precise calculations and the need to consistently apply the formula. By the end of this guide, you'll have a thorough comprehension of the mass percent equation, and you'll be able to confidently solve related problems.
Throughout this article, we'll explore the intricacies of the mass percent equation, provide detailed solutions to common problems, and highlight how this equation is applied in real-world scenarios. Whether you're preparing for an exam, completing a homework lesson on equations for proportional relationships, or simply looking to enhance your mathematical skills, this guide will serve as a valuable resource. Let's dive in and demystify the mass percent equation.
Step-by-Step: How to Solve Mass Percent Equation
Step 1: Identifying the Components
The first step in using the mass percent equation is to clearly identify the component whose mass percent you want to calculate. This involves understanding the problem statement and determining the masses of each component involved. For example, if you're given a solution of saltwater, you need to know the mass of the salt and the mass of the water. Accurate identification of these components is essential to avoid errors later in the calculation.
Step 2: Calculating the Total Mass
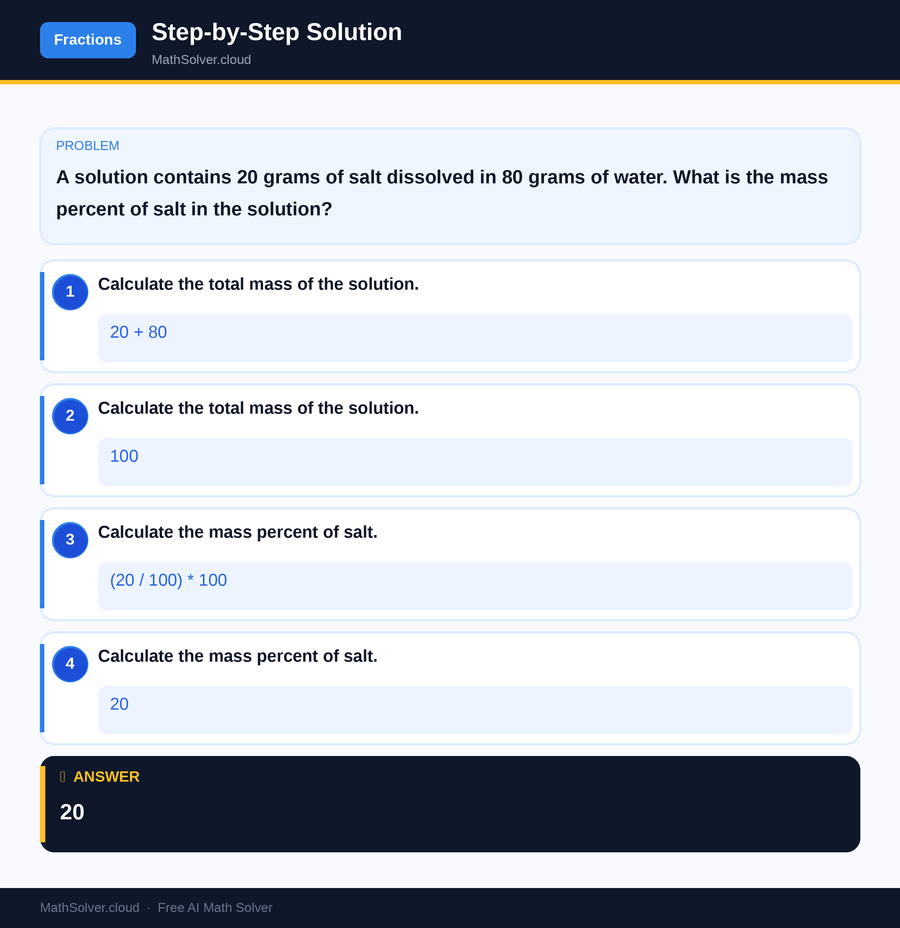
Once you've identified the components, the next step is to calculate the total mass of the mixture. This is done by summing the masses of all individual components. Continuing with our saltwater example, if you have 20 grams of salt and 80 grams of water, the total mass of the solution would be 20 grams + 80 grams = 100 grams. Ensuring that you have the correct total mass is crucial for the accuracy of the mass percent equation.
Step 3: Applying the Mass Percent Equation
With the component's mass and the total mass in hand, you can now apply the mass percent equation. Using the formula Mass percent = (mass of component / total mass of mixture) x 100, substitute the known values to find the mass percent. This step involves basic arithmetic but requires attention to detail to ensure correct substitution and calculation.
Step 4: Expressing the Result
Finally, after calculating the mass percent using the equation, you need to express the result clearly, typically rounding to the appropriate number of decimal places. This final step is important as it communicates the concentration of the component in a clear and precise manner, which is essential for both academic and practical applications.
🤖 Stuck on a math problem?
Take a screenshot and let our AI solve it step-by-step in seconds
⚡ Try MathSolver Free →Worked Examples
Example 1
MathSolver Chrome extension solving this problem step-by-step
Example 2
MathSolver Chrome extension solving this problem step-by-step
Common Mistakes to Avoid
One common mistake students make with the mass percent equation is misidentifying the components or inaccurately calculating the total mass of the mixture. This often leads to incorrect calculations and results. To avoid this, always double-check the values provided in the problem statement and ensure that you're summing the correct masses.
Another frequent error is neglecting units and failing to convert them when necessary. Mass percent calculations require consistent units throughout the process. Ensure that all masses are in the same unit (e.g., grams) before performing any calculations. This attention to detail will help you avoid unnecessary mistakes and ensure accurate results.
Real-World Applications
The mass percent equation is widely used in various real-world applications, particularly in chemistry and material science. For example, it is crucial in calculating the concentration of solutions in pharmaceutical formulations, where precise amounts of active ingredients are necessary to ensure efficacy and safety.
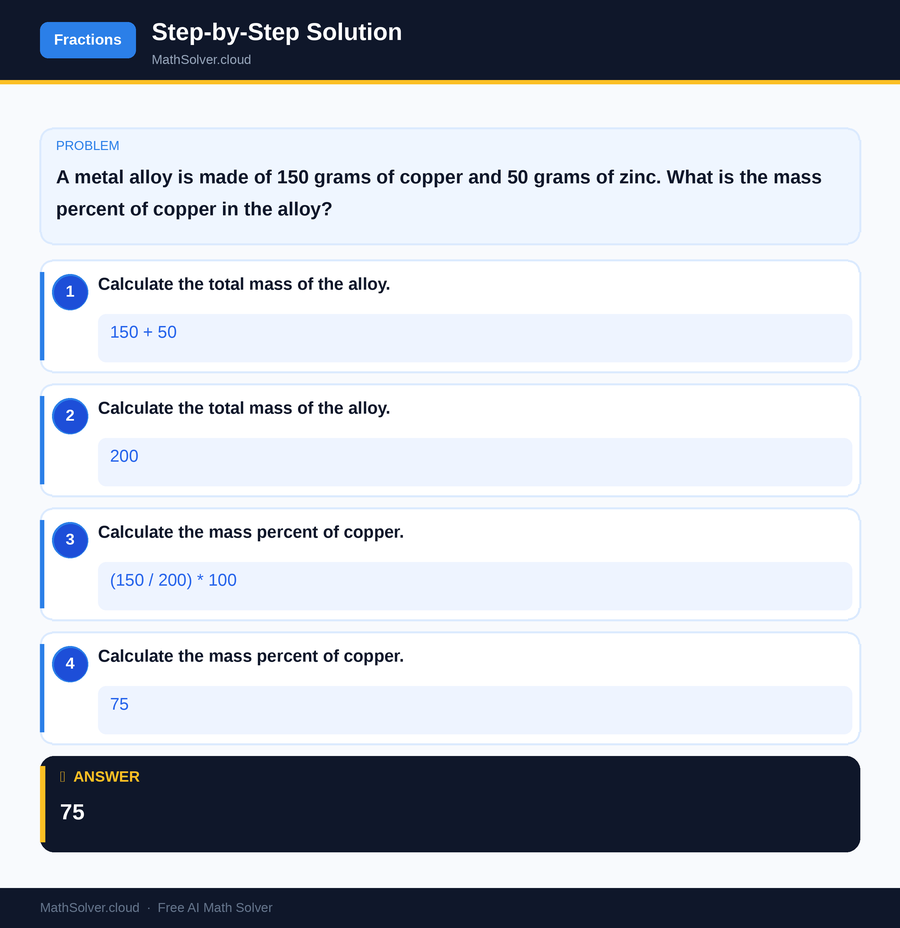
Moreover, the mass percent equation is employed in industries that manufacture alloys, such as those used in construction or electronics. Understanding the composition of an alloy, for example, the proportion of copper or zinc, is essential for determining its properties and suitability for specific applications. These examples highlight the importance of mastering the mass percent equation, as it plays a vital role in both academic and practical scenarios.
Frequently Asked Questions
❓ What is the mass percent equation and how is it used?
❓ Why do students often struggle with the mass percent equation?
❓ How can AI help with the mass percent equation?
❓ What is the difference between the mass percent equation and other types of equations?
❓ How is the mass percent equation relevant in everyday life?
🚀 Solve any math problem instantly
2,000+ students use MathSolver every day — join them for free
📥 Add to Chrome — It's Free