Mastering the Chemical Equation Calculator
📋 In this guide
A chemical equation calculator is a powerful tool designed to assist students and professionals in solving complex chemical reactions by analyzing and balancing chemical equations. Often, students find themselves struggling with the intricacies of chemistry, especially when it comes to balancing chemical equations. This can be due to the abstract nature of chemical symbols and the precision required to achieve a balanced equation. With the help of a chemical equation calculator, the process becomes more manageable, allowing students to focus on understanding the underlying concepts rather than getting lost in the arithmetic.
In chemistry, balancing chemical equations is essential because it ensures that the Law of Conservation of Mass is upheld. This law states that matter cannot be created or destroyed in a closed system, so the same number of each type of atom must exist on both sides of the equation. However, achieving this balance can be challenging, particularly for beginners who are still familiarizing themselves with chemical symbols and stoichiometry. A chemical equation calculator simplifies this process by providing instant solutions, enabling students to verify their manual attempts or learn from a step-by-step breakdown of the solution.
By the end of this article, you will gain a comprehensive understanding of how to use a chemical equation calculator effectively. You will learn about key formulas, step-by-step instructions, common mistakes to avoid, and the real-world applications of these calculators. Additionally, you'll find answers to frequently asked questions that will further enhance your understanding and ability to apply these tools in your studies or professional work.
Step-by-Step: How to Solve Chemical Equation Calculator
Step 1: Understand the Chemical Equation
Before using a chemical equation calculator, it is crucial to understand the components of the chemical equation you are working with. Identify the reactants and products involved in the reaction. Write down the unbalanced equation, ensuring you have the correct chemical formulas for each compound. For instance, in the reaction of hydrogen and oxygen to form water, the unbalanced equation is H2 + O2 -> H2O. Familiarize yourself with the atomic masses of the elements involved, as this information is vital for further calculations.
Step 2: Identify the Atoms and Their Count
Once you have the unbalanced equation, the next step is to identify all the different types of atoms present in the equation and count the number of each atom type on both sides. This step helps in determining how unbalanced the equation is and guides the process of balancing. For example, in the reaction H2 + O2 -> H2O, you have two hydrogen atoms and two oxygen atoms as reactants, but the product side only has two hydrogen atoms and one oxygen atom.
Step 3: Use the Chemical Equation Calculator
Input the unbalanced chemical equation into the chemical equation calculator. Depending on its capabilities, the calculator will provide a balanced equation by adjusting the coefficients of each compound. For example, a balance chemical equation calculator might take the unbalanced equation H2 + O2 -> H2O and return the balanced equation 2H2 + O2 -> 2H2O. This process ensures that the number of each type of atom is equal on both sides, maintaining the Law of Conservation of Mass.
Step 4: Verify and Understand the Solution
After obtaining the balanced equation, it is essential to verify the result manually to ensure full understanding. Count the atoms of each element on both sides to confirm the equation is balanced. This step helps reinforce learning and ensures that you can balance equations independently in the future. Additionally, if the chemical equation calculator provides detailed steps, review them to understand the logic behind each adjustment made.
🤖 Stuck on a math problem?
Take a screenshot and let our AI solve it step-by-step in seconds
⚡ Try MathSolver Free →Worked Examples
Example 1
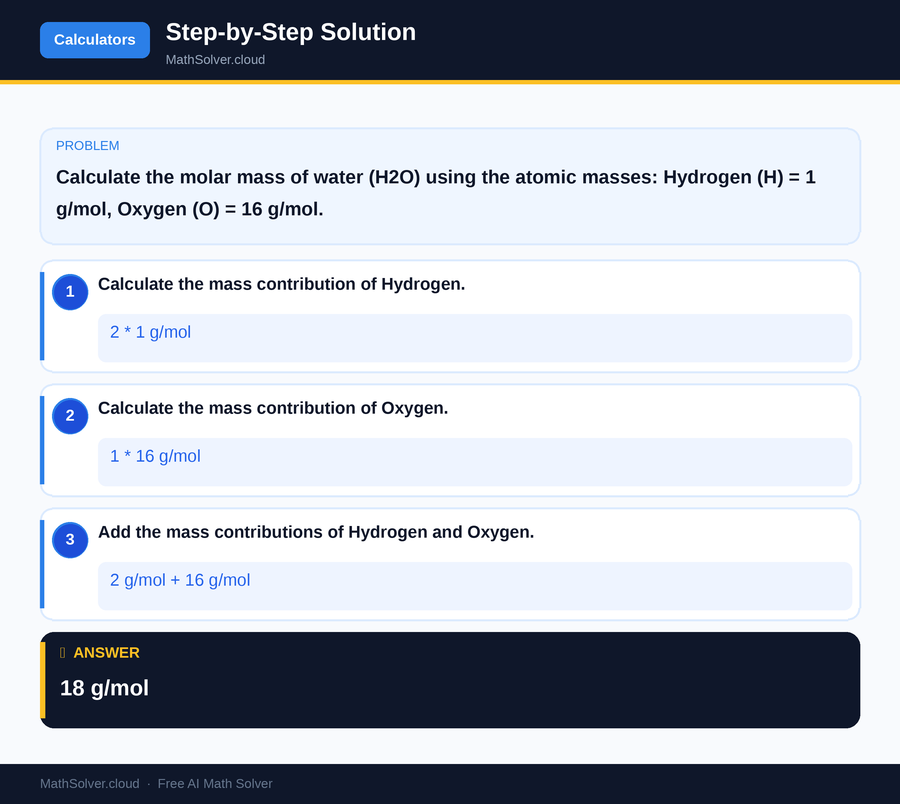
MathSolver Chrome extension solving this problem step-by-step
Example 2
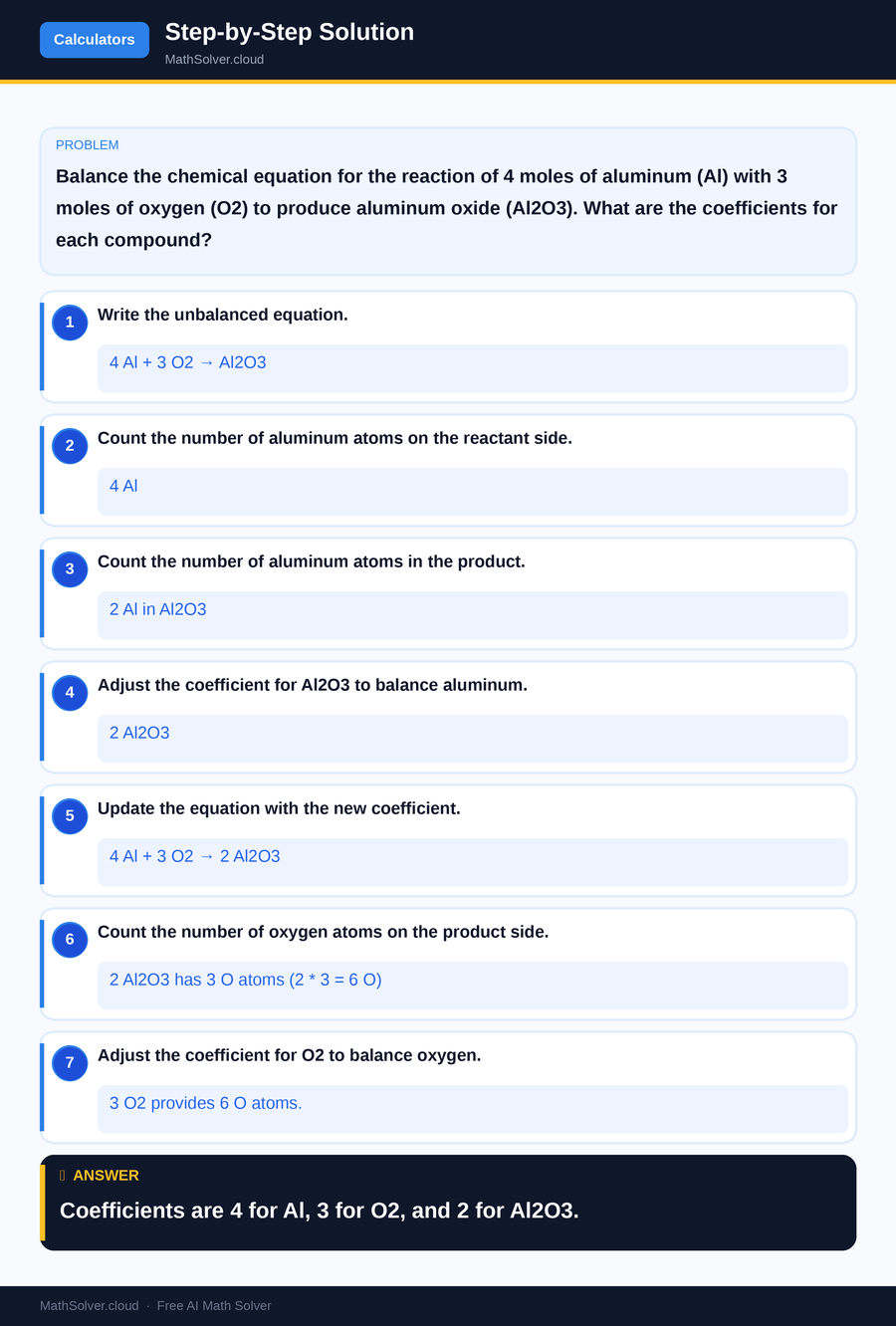
MathSolver Chrome extension solving this problem step-by-step
Common Mistakes to Avoid
One common mistake students make is altering the subscripts of chemical formulas instead of adjusting the coefficients. Changing subscripts changes the actual compound, which is incorrect. Always adjust coefficients to balance the equation. Another mistake is neglecting to verify the balance by recounting atoms on each side. This step is crucial to ensure the equation truly adheres to the Law of Conservation of Mass. Avoid these pitfalls by consistently checking your work and using a reliable balancing chemical equation calculator with steps to guide you.
Real-World Applications
Chemical equation calculators are invaluable in various real-world applications. In the pharmaceutical industry, precise chemical reactions are necessary for drug formulation, where balanced equations ensure correct proportions of reactants. Environmental science also relies on chemical equations to model pollutant interactions and their impact on ecosystems. Additionally, educators and students use these calculators to facilitate learning, with tools like our complete Online Calculators guide providing comprehensive resources for mastering chemistry concepts.
Frequently Asked Questions
❓ What is a chemical equation calculator and how does it work?
❓ Why is balancing chemical equations important in chemistry?
❓ How can AI help with using a chemical equation calculator?
❓ Can a chemical equation calculator handle complex reactions with multiple products?
❓ How are chemical equation calculators used in educational settings?
🚀 Solve any math problem instantly
2,000+ students use MathSolver every day — join them for free
📥 Add to Chrome — It's Free