Balanced Chemical Equation Calculator: A Comprehensive Guide
📋 In this guide
A balanced chemical equation calculator is a powerful tool designed to help students and professionals alike in the task of balancing chemical equations. Balancing chemical equations is an essential skill in chemistry, yet it can be challenging for many students due to the complexity involved in ensuring that the same number of each type of atom appears on both sides of the equation. In this article, you'll learn about the intricacies of using a balanced chemical equation calculator, the step-by-step process of balancing equations, and how this tool can simplify your chemistry studies.
Students often struggle with balancing chemical equations because it requires a firm understanding of stoichiometry and the ability to manipulate algebraic expressions. This challenge can be compounded by the introduction of more complex reactions involving multiple reactants and products. By using a balanced chemical equation calculator, students can gain confidence in their ability to balance equations accurately and efficiently, thereby enhancing their overall understanding of chemistry.
In this guide, you will learn how to use a balanced chemical equation calculator effectively, with detailed examples and explanations provided to illustrate the process. Whether you are learning the basics or tackling more advanced chemistry problems, this tool can serve as a valuable resource in your educational journey. Additionally, we'll discuss common mistakes to avoid, real-world applications, and address frequently asked questions related to using this calculator.
Step-by-Step: How to Solve Balanced Chemical Equation Calculator
Step 1: Identify the Reactants and Products
The first step in using a balanced chemical equation calculator is to clearly identify all the reactants and products involved in the chemical reaction. Reactants are the substances that start a chemical reaction, while products are the substances formed as a result of the reaction. For example, in the combustion of propane, the reactants are propane (C3H8) and oxygen (O2), and the products are carbon dioxide (CO2) and water (H2O).
Step 2: Write the Unbalanced Equation
Once you have identified the reactants and products, write the unbalanced equation. This involves listing the chemical formulas of the reactants on the left side of the arrow and the chemical formulas of the products on the right side. Using the previous example, the unbalanced equation for the combustion of propane is: C3H8 + O2 -> CO2 + H2O.
Step 3: Count the Atoms for Each Element
The next step is to count the number of atoms for each element present in both the reactants and the products. This involves breaking down each chemical formula into its constituent elements and tallying the atoms. For instance, in the equation C3H8 + O2 -> CO2 + H2O, you would count the number of carbon, hydrogen, and oxygen atoms on each side of the equation.
Step 4: Balance the Equation
Finally, use coefficients to balance the equation so that the number of atoms for each element is equal on both sides. Coefficients are numbers placed before the chemical formulas to indicate the number of molecules or moles involved. Adjust these coefficients systematically, starting with the most complex molecule, until the equation is balanced. This may require some trial and error, but a balanced chemical equation calculator can simplify this process by providing a step-by-step solution.
🤖 Stuck on a math problem?
Take a screenshot and let our AI solve it step-by-step in seconds
⚡ Try MathSolver Free →Worked Examples
Example 1

MathSolver Chrome extension solving this problem step-by-step
Example 2
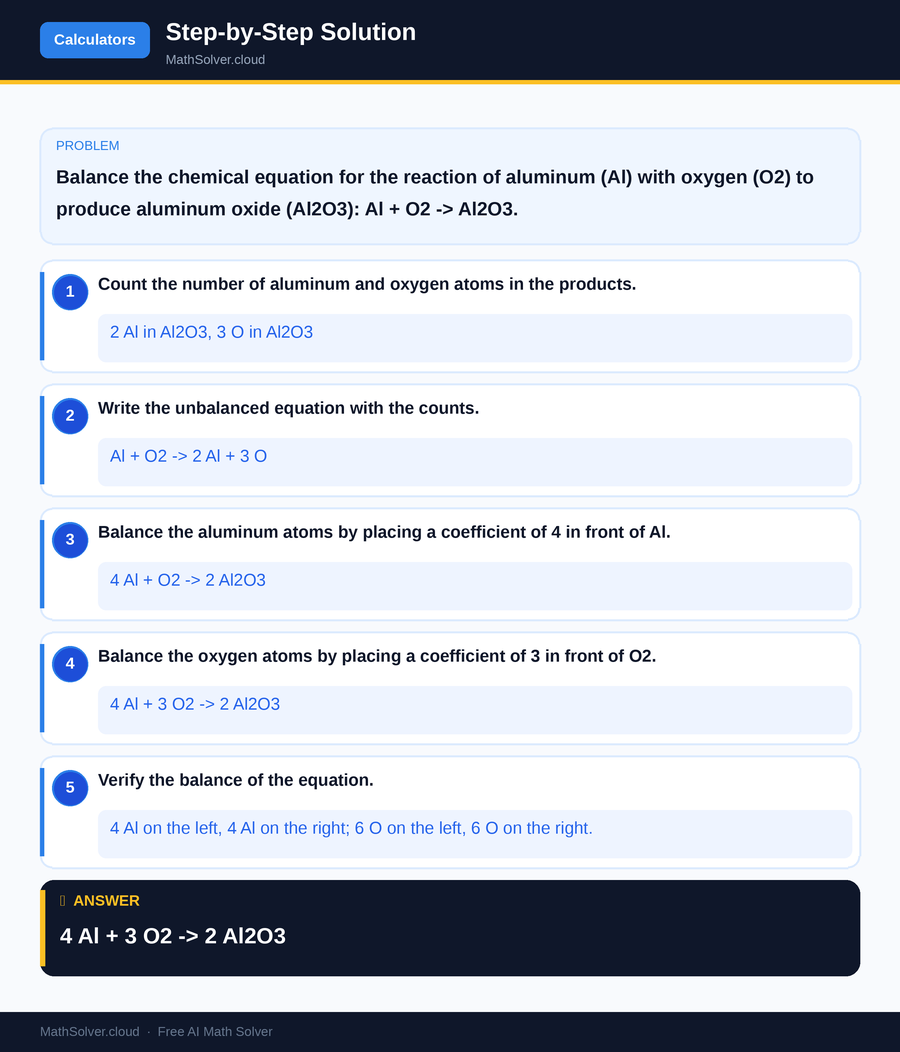
MathSolver Chrome extension solving this problem step-by-step
Common Mistakes to Avoid
One of the most common mistakes students make when balancing chemical equations is not properly counting the atoms of each element. This can lead to errors in balancing and incorrect coefficients. To avoid this, always double-check your atom counts on both sides of the equation before adjusting coefficients.
Another frequent error is attempting to balance equations by changing subscripts instead of coefficients. Subscripts are part of the chemical identity of the compound and should not be altered. Instead, use coefficients to balance the equation, as these indicate the number of molecules or moles involved in the reaction.
Real-World Applications
Balanced chemical equation calculators are used extensively in various fields, such as chemistry research, pharmaceuticals, and environmental science. For instance, in the pharmaceutical industry, accurate chemical equations are crucial for formulating medications and ensuring the correct proportions of ingredients.
In environmental science, balanced equations are used to model chemical reactions in the atmosphere, such as the formation of ozone or the combustion of fossil fuels. Understanding these reactions helps scientists develop strategies to reduce pollution and mitigate climate change.
Frequently Asked Questions
❓ What is a balanced chemical equation calculator and how does it work?
❓ Why is balancing chemical equations important?
❓ How can AI help with balanced chemical equation calculators?
❓ What other tools can help with solving chemistry problems?
❓ How can balanced chemical equation calculators be practically used in studies?
🚀 Solve any math problem instantly
2,000+ students use MathSolver every day — join them for free
📥 Add to Chrome — It's Free